Habitat Mapping
Much of the research work that Ocean Ecology was involved in could be described as habitat mapping. When habitat mapping is carried out in the marine environment, the product is generally referred to as a “benthic habitat map” – a “spatial representation of physically distinct areas of seafloor that are associated with particular groups of plants and animals.” (Harris and Baker. 2012. Seafloor geomorphology as benthic habitat GeoHAB atlas of seafloor geomorphic features and benthic habitats).
The term benthic refers to anything associated with or occurring on the bottom of a body of water. The animals and plants that live on or in the bottom are known as the benthos.
In ocean waters, nearshore and estuary areas are most frequently mapped. This is due in part to the fact that these areas are shallow enough to map, but also because these areas are very important conservation targets.
Benthic habitat maps are derived from aerial imagery, underwater photos, acoustic surveys, and data gathered from sediment samples. The resulting digital map is viewed using geographic information system tools.
Benthic habitat maps provide:
- a summary of the physical and biological resources of a region
- a map of species distribution and abundance.
- information on water and habitat quality.
- a means to monitor changing ecological communities and document invasive species.
- data on fisheries resources.
Benthic habitat maps are important because they:
- support informed decisions about managing our coastlines.
- provide data for long-term monitoring.
- form a baseline for assessing future anthropogenic impacts.
- identify critical and sensitive habitats.
Towed Benthic Video Camera
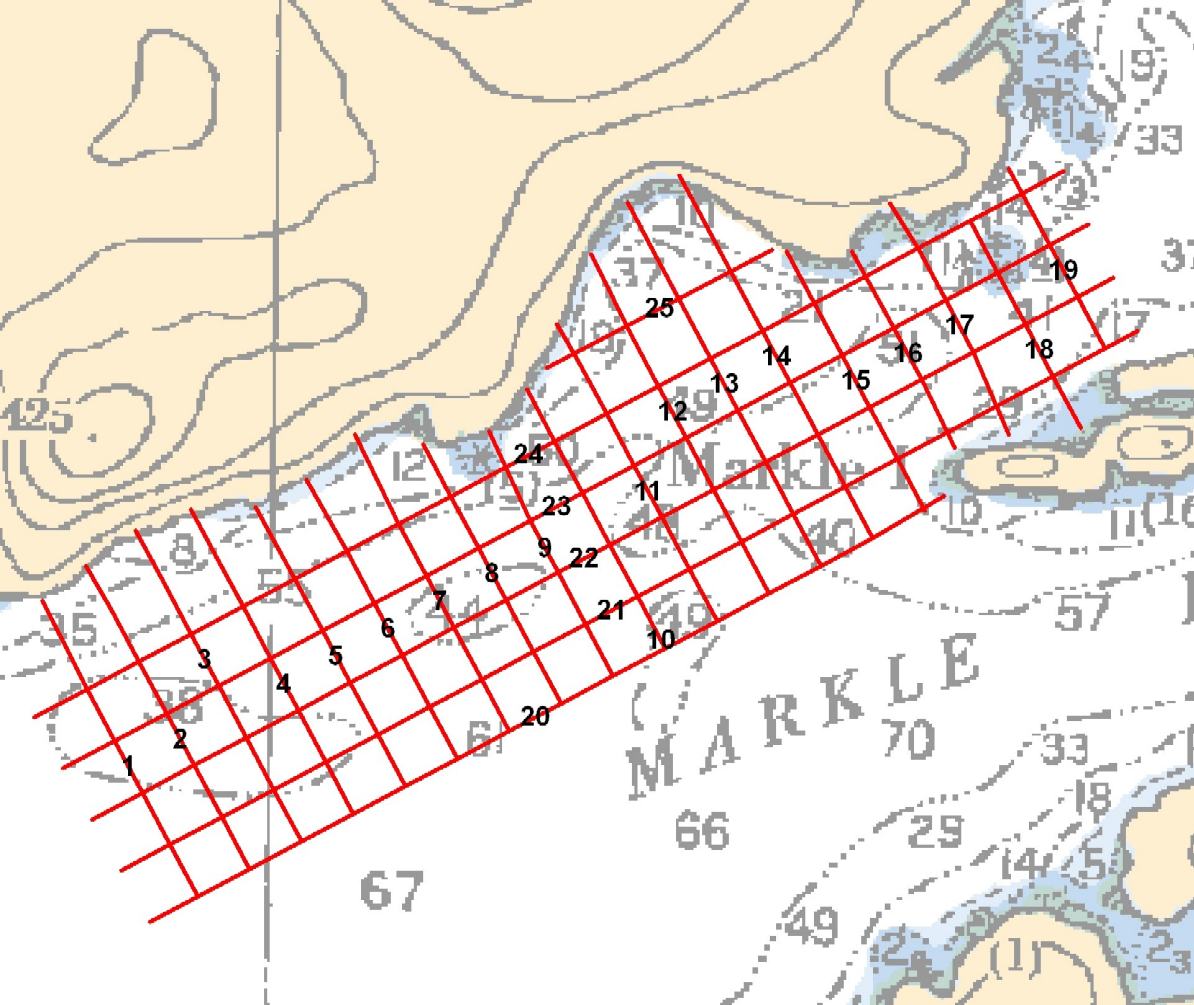
Much of Ocean Ecology’s benthic habitat mapping was carried out using a GPS-positioned, towed video camera, called the “Benthic Video Surveyor 2“, to collect imagery of the seabed. Typically, this camera was towed approximately 1 m above the seabed, generating a video image which was stamped with the GPS position (latitude/longitude) and the date and time. High intensity LEDs were mounted on the camera to provide illumination when required. For more information on this system, refer to our paper Chatham Sound Eelgrass Study Final Report.
We usually carried out our benthic video surveys as grid transects (see image at the top of this post), with nominal shore-normal and shore-parallel transect line spacing ranging between 25 m and 100 m, depending on the degree of resolution desired. The depths of the surveys ranged from 5.0 m to approximately 150 m.
Examples of some of our video footage are shown below.
Transect above a sea pen bed
Video footage taken by the benthic video system showing abundant life in a rocky subtidal zone
We also extracted still images from the video footage to get some interesting shots of marine life.




Video Signal Processing
The analog camera signals from any of our benthic video camera systems could be recorded directly onto a hard drive by a computer using the open source software iSpy and two usb video capture devices. The live video signal could also be viewed directly on the computer monitor as we were doing the transects. For the purposes of video analyses when a dual camera system was used, the forward-looking and downward-looking videos were combined into a 2 x 2 quad format, with the forward-looking camera in the upper left position and the downward-looking camera in the lower right position (as seen in the video footage shown above).

Video Annotation
Once video footage from a benthic video survey had been collected, it was viewed and annotated. The annotation process consisted of coding each second of raw video for types and percentage composition of substrate, species of fauna presence and their abundances, and species of flora presence and their percentage coverage.
There are two main approaches to video annotation – frame annotation and timeline annotation. Depending on the particular type of analyses that needed to be performed, we used both frame annotation (e.g., using VARS [Video Annotation and Reference System] developed by the Monterey Bay Aquarium Research Institute [MBARI]) and timeline annotation (e.g., using Anvil developed by Michael Kipp of the German Research Center for Artificial Intelligence [DFKI]).
Frame Annotation
In frame annotation, each frame of the video can be annotated individually. This generates a series of unique data points, one for each video frame. This is very useful for statistical analyses, but can be very tedious and repetitous from the annotation perspective, as often a single annotation event (e.g., the presence of a particular patch of algae or the movement of an individual fish) may span a number of frames.
Shown below is an example of a video being annotated using frame annotation.

Timeline Annotation
In timeline annotation, segments of the video timeline can be annotated for each “event”. This generates a start and end time for each observed event. This type of annotation is much more rapid than frame annotation; however, the type of data generated by timeline annotation is much more difficult to analyze statistically, and must be transformed into point data for most statistical work.
Shown below is an example of a video being annotated using timeline annotation.

Annotation Analyses
All annotation data were entered into a relational database, either Microsoft Access or Apache Derby. The database could then be queried for specific data records, or used for further statistical analyses on the data. Data from the database could also be imported into GIS applications for mapping.
Transect Video Replay Software
Since all the recorded video was georeferenced using the ship’s onboard GPS system, it was possible to view the video in spatial relationship to a chart of the study site. Using transect video replay software, a specific position on a particular transect could be selected, and the video footage corresponding to that location could then be viewed. The position marker on the map moves synchronously with the video replay, thus showing the exact location of each video frame on the map.
Ocean Ecology used several versions of transect video replay software, depending on the project and data viewing requirements.
Standalone Java-based Viewer
We wrote our own in-house java-based software for viewing transect video data. This viewer was portable and could be used on a DVD or USB flash drive. It was standalone, and did not require an internet connection for data download.

ActionCam and MapServer
Alternately, we used ActionCam, a GPS video viewer which can overlay GPS data on a variety of background maps provided by internet servers. Therefore, this system required an internet connection. A portable version of MapServer was used to serve local project data to ActionCam.

Mapping Biodiversity
Biodiversity is the variation of life forms within a given ecosystem, biome, or for the entire Earth. Biodiversity is often used as a measure of the health of biological systems.
Once we had produced the range maps for the all the species found at a particular site (see our page on species distribution maps), we were able to use the range map polygons to calculate the biodiversity of the site.
Biodiversity Indices
Some of the biodiversity indices which we calculated were:
- Shannon’s diversity index – the Shannon’s diversity index is an “information index” which measures biodiversity based on the principle that diversity can be measured much like the information contained in a code or message (e.g., the longer and more complex the code, the greater the diversity). The Shannon’s diversity index is affected by both the number of species and their equitability, or evenness. A greater number of species and a more even distribution of species both result in an increase in Shannon’s diversity. The maximum Shannon’s diversity for a sample is found when all species are equally abundant. Values of the Shannon’s diversity index for real communities typically fall between 1.5 and 3.5.
- Shannon’s evenness – Shannon’s evenness is derived from Shannon’s diversity index. Evenness is a measure of how similar the abundances of different species are. When there are similar proportions of all species, evenness approaches a value of 1.0. When the abundances are very dissimilar (some rare and some common species), then the value for evenness decreases.
- Simpson’s dominance index – the Simpson’s dominance index measures biodiversity based on the probability that two individuals randomly selected from a sample will belong to the same species (or some category other than species). Simpson’s dominance index ranges from 0 (all taxa are equally present) to 1.0 (one taxon dominates the community completely).
- Species richness – species richness is the simplest measure of biodiversity, and is simply a count of the number of different species in a given area. Species richness is also referred to as alpha-diversity. Species richness is commonly used, along with other factors, as a measure for determining the overall health of different biological ecosystems. High species richness for a given area indicates a high level of ecosystem stability, thus allowing the ecosystem to better withstand natural or anthropogenic disturbances (i.e. fires, floods, disease, deforestation, etc.). Therefore, high levels of species richness in ecosystems typically characterize these ecosystems as healthy and robust.
Biodiversity Analysis Example

Shown above is an example of the variation of species richness throughout a site. The bottom map shows all the species range polygons overlaid on one another. The top map shows the species richness for the site based on the range polygons. Red indicates areas of high species richness, whereas green indicates areas of low species richness.
The following values were calculated for this site:
- Species richness = 16
- Shannon’s diversity index = 2.44
- Shannon’s evenness = 0.88
- Simpson’s dominance index = 0.30
These values indicate that this site has a relatively even distribution of species and a moderate level of biodiversity.
For a real life example of a biodiversity analysis of a site, see our paper Appendix D: Towed Benthic Video Survey of Site 1 for the Canpotex Potash Terminal Project Disposal at Sea Application. In Proposed New Disposal at Sea Sites For Canpotex Potash Export Terminal, Ridley Island, Prince Rupert, BC.
High Definition Drop Camera
The High Definition Drop Camera system was designed to take high resolution images of the seafloor for analytical work. The camera was mounted in a drop frame to reduce blur from camera motion and ensure that all images were taken at the same height above the seafloor and had the same field of view.
Shown below is an example of a photo of eelgrass taken by the high definition drop camera.

For further details on the high definition drop camera and examples of how we deployed it, see our papers Experimental Herring Video Survey Final Report and Lucy Islands Eelgrass Study.
Deep Lander
The Deep Lander was a drop video camera system designed specifically for taking video footage of the seafloor in deep water environments.


For further details on the deep lander and examples of how we deployed it, see our papers Appendix E: Drop Camera Video Survey of Site 2 for the Canpotex Potash Terminal Project Disposal at Sea Application. In Proposed New Disposal at Sea Sites For Canpotex Potash Export Terminal, Ridley Island, Prince Rupert, BC and Lucy Islands Eelgrass Study.
Benthic Macroinvertebrate Analysis
Often, when we deployed the deep lander, we also collected samples of the marine sediment for benthic macroinvertebrate analysis in order to get a better picture of the types and numbers of organisms inhabiting the benthos.

Marine benthic macroinvertebrates were collected using a grab sampler or corer. The sediment sample was then screened to remove fine-grained sediment, and the macroinvertebrates were hand sorted from the remaining coarse material. The animals were preserved, and later examined under a magnifying lens or dissecting microscope. Organisms were enumerated and identified to the genus, and in many cases, species level.

For an examples of a macroinvertebrate analysis, see our paper Appendix C: Macroinvertebrate Baseline Study for the Canpotex Potash Terminal Project Disposal at Sea Application. In Proposed New Disposal at Sea Sites For Canpotex Potash Export Terminal, Ridley Island, Prince Rupert, BC.
Beach Seining
Another way of observing and recording biodiversity in the nearshore environment is beach seining. Beach seining is an efficient method to capture salmonids and some nonsalmonid fishes in a wide variety of habitats, including rivers, estuarine, and nearshore lake, reservoir, and marine habitats. It is most effective when used in relatively shallow water with few obstructions, where fish are in high concentrations, and for species that are less likely to outswim the net. Seining permits the capture and release of fish without significant stress or harm. In our studies, we have carried out beach seine surveys using a 30 foot minnow seine with a 1/8″ mesh following the protocol set out in the Salmonid Field Protocols Handbook.
Fish caught in the beach seine are identified, measured (length and weight), photographed, and released.

For an example of a beach seine analysis, see our paper Lucy Islands Eelgrass Study.
Shoreline Surveys
Although the nearshore and intertidal habitats are often surveyed using very different techniques, these two environments form a continuum, and should be considered together in order to provide a full understanding of the marine ecosystem at a particular location.
Working as Ocean Ecology, we have carried out both quantitative (e.g., stratified vertical quadrat surveys or random sampling along a horizontal transect, and identification and enumeration of intertidal organisms; see Marine Foreshore Environmental Assessment Procedures, Physical Shore-Zone Mapping System for British Columbia, and Coastal/Estuarine Fish Habitat Description and Assessment Manual, Part II, Habitat Description Procedures) and qualitative (e.g., photodocumentation; see Photo Point Monitoring Protocols and Measuring Aea/Length of Organisms with GIS) shoreline surveys. Handheld GPS units were used for georeferencing of data.


We often recorded data using android tablets and survey software, such as CyberTracker, and later uploaded the data into a GIS program for further mapping and data analyses.